PDF) Thermo physical properties of acetone–zinc bromide for using in a low temperature driven absorption refrigeration machine | Salman Ajib and Ali Karno - Academia.edu
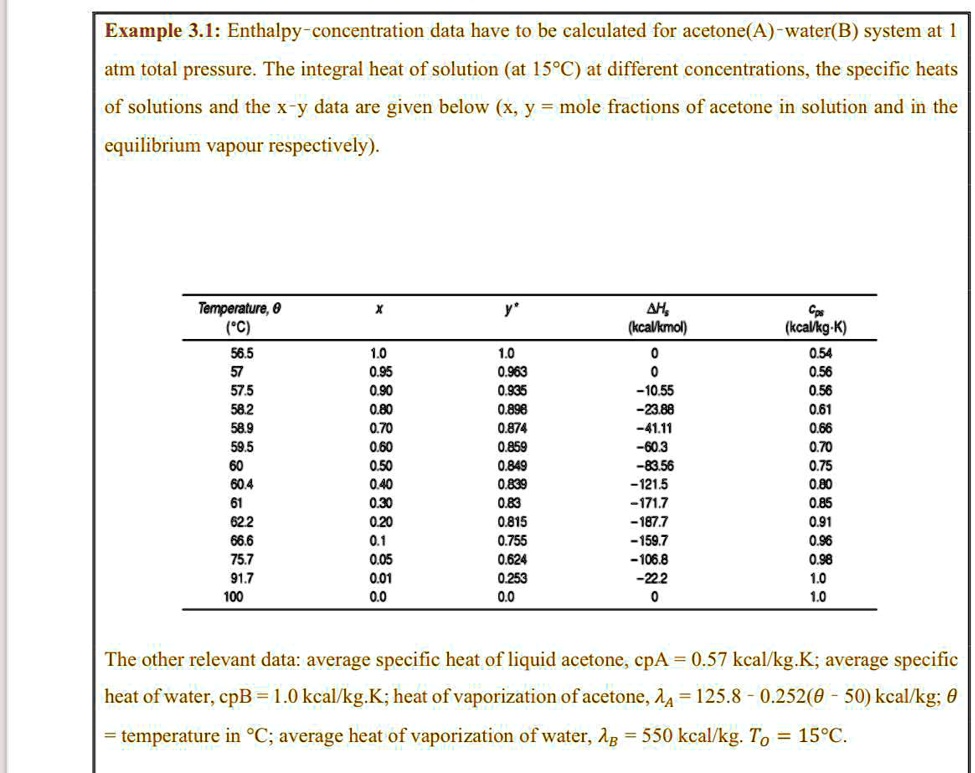
SOLVED: Example 3.1: Enthalpy-concentration data have to be calculated for acetone-water system at 1 atm total pressure. The integral heat of solution at 15°C at different concentrations, the specific heats of solutions,
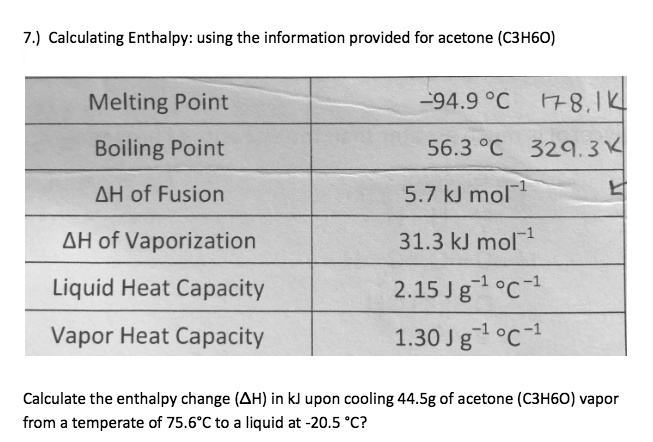
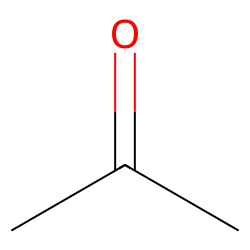
SOLVED: Calculating Enthalpy: using the information provided for acetone (C3H6O) Melting Point Boiling Point AH of Fusion 94.9 °C 56.3 °C 329.34 kJ mol-1 AH of Vaporization 31.3 kJ mol-1 2.15 J

Estimation of Diffusion Coefficients in Acetone−Cellulose Acetate Solutions | Industrial & Engineering Chemistry Research

Acetone heat capacities: (c L and c G ) for the molecular liquid and... | Download Scientific Diagram
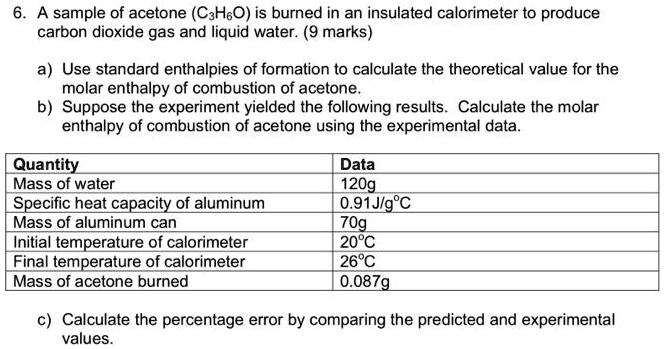
SOLVED: A sample of acetone (C3H6O) is burned in an insulated calorimeter to produce carbon dioxide gas and liquid water. Use standard enthalpies of formation to calculate the theoretical value for the

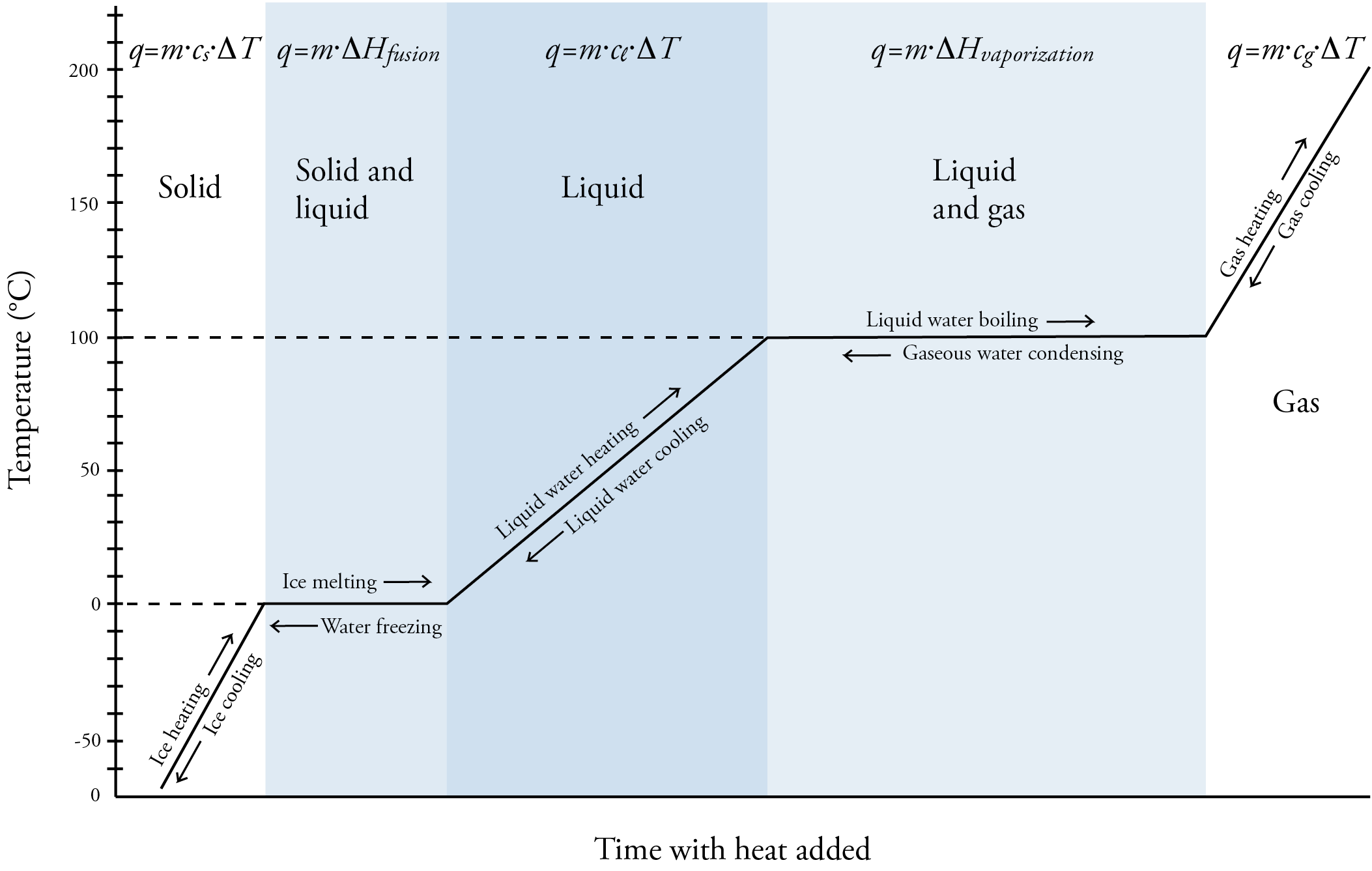
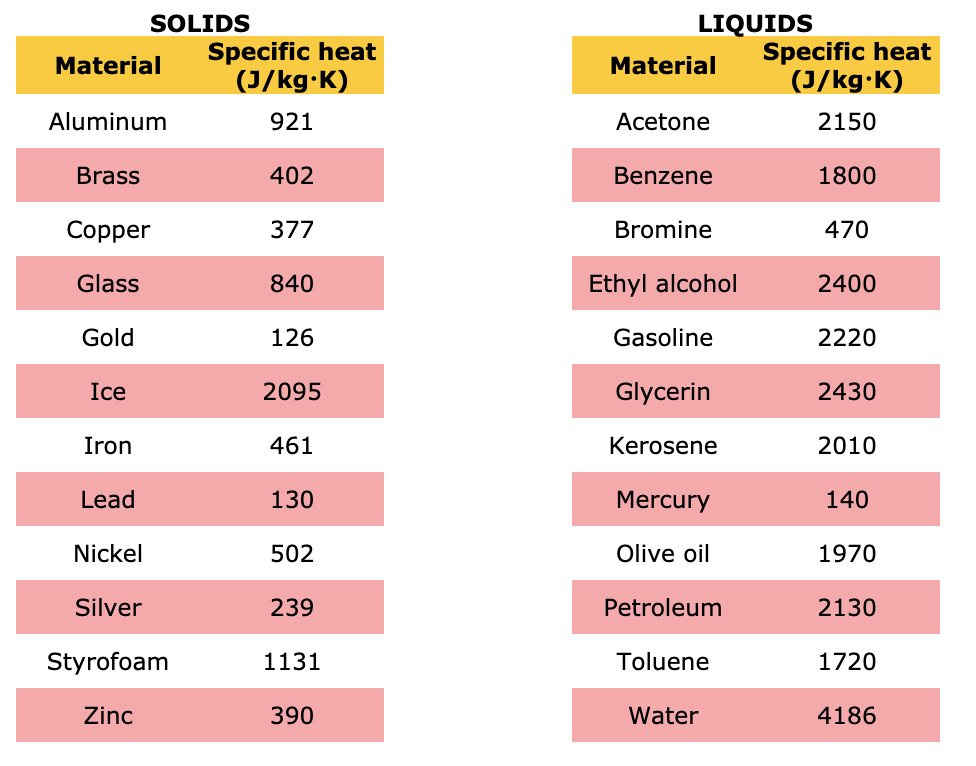
Laboratory Manual: Bachelor of Engineering Technology Chemistry For Engineering Technologist | PDF | Evaporation | Liquids
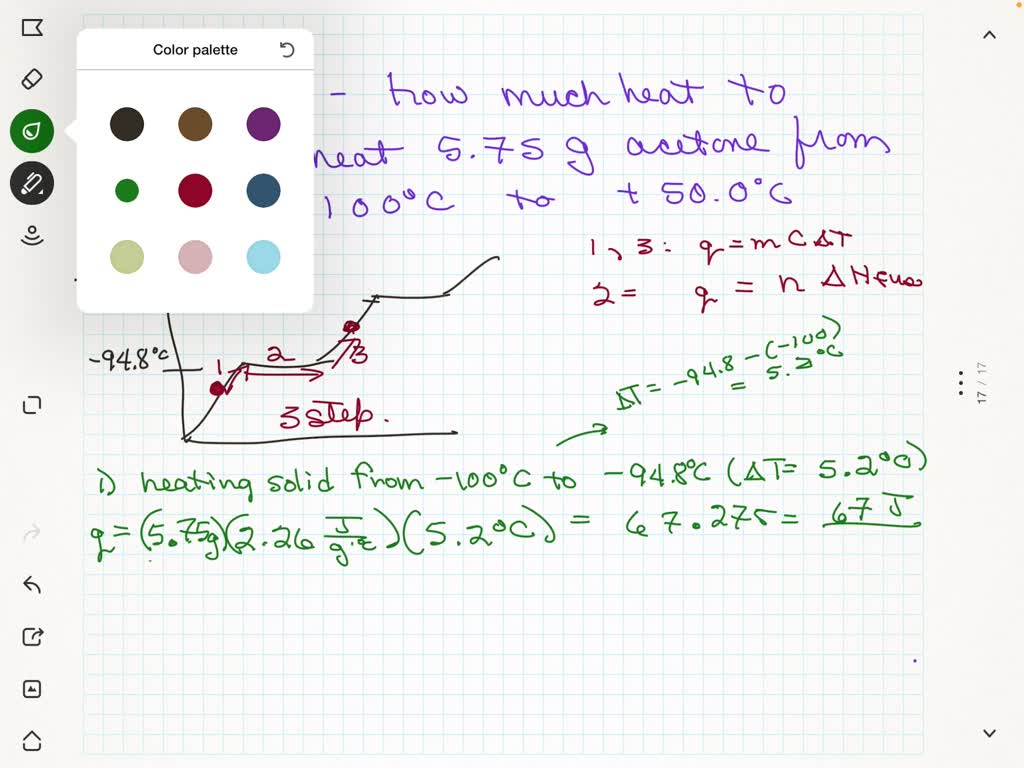
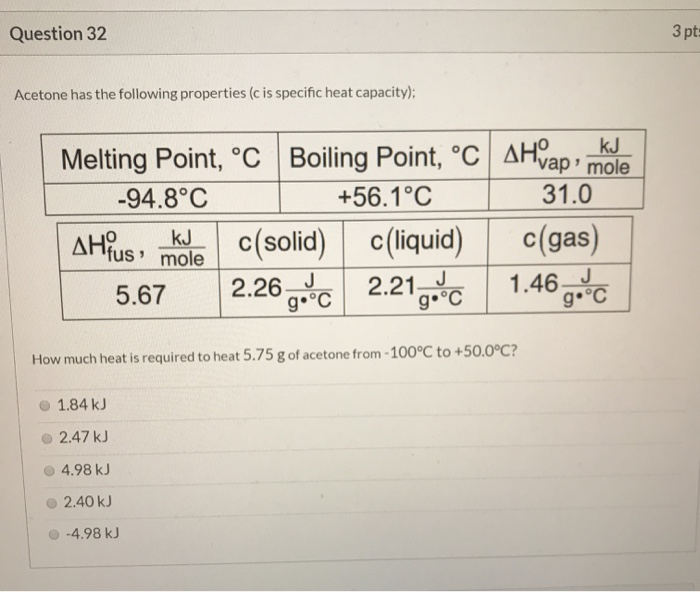
SOLVED: Acetone has the following properties (c is specific heat capacity): Melting Point: -94.8°C Boiling Point: +56.1°C AHYap: 31.0 kJ mole (solid) AH?s: 5.67 kJ mole (liquid) c(gas): 2.26 g**C How much

Thermophysical Properties of Liquid Acetone, Cellulose Acetate, and... | Download Scientific Diagram